Slide 1 - Zinc-Copper Cell Example Cell Potential & Notation
Zinc-Copper Cell Example Cell Potential & Notation
Notes on Electrochemical Principles
---
Photo by Stephan HK on Unsplash

Generated from prompt:
Write notes on galvanic cell, using zinc-copper cell as example. Determining the cell potential and cell notation
This presentation introduces galvanic cells, which generate electricity from spontaneous redox reactions. Using the classic zinc-copper cell as an example, it covers cell components, half-cell reactions (Zn oxidation at anode, Cu reduction at cathode), cell notation (Zn | Zn²⁺ || Cu²⁺ | Cu), standard reduction potentials, and cell potential calculation (E°_cell = 1.10 V). Includes agenda, diagrams, and key takeaways on electrochemical principles foundational to batteries.
Zinc-Copper Cell Example Cell Potential & Notation
Notes on Electrochemical Principles
---
Photo by Stephan HK on Unsplash

---
Photo by Alfred Quartey on Unsplash

1
Generates electricity from spontaneous redox reactions
---
Photo by Ryan Zazueta on Unsplash

Source: Wikipedia: Galvanic cell


---
Photo by Claudio Schwarz on Unsplash

Anode (Oxidation) Zn(s) → Zn²⁺(aq) + 2e⁻ Less noble metal (Zn) loses electrons
Cathode (Reduction) Cu²⁺(aq) + 2e⁻ → Cu(s) More noble metal (Cu) gains electrons


4
Using standard electrode potentials
---
Photo by Thorium on Unsplash

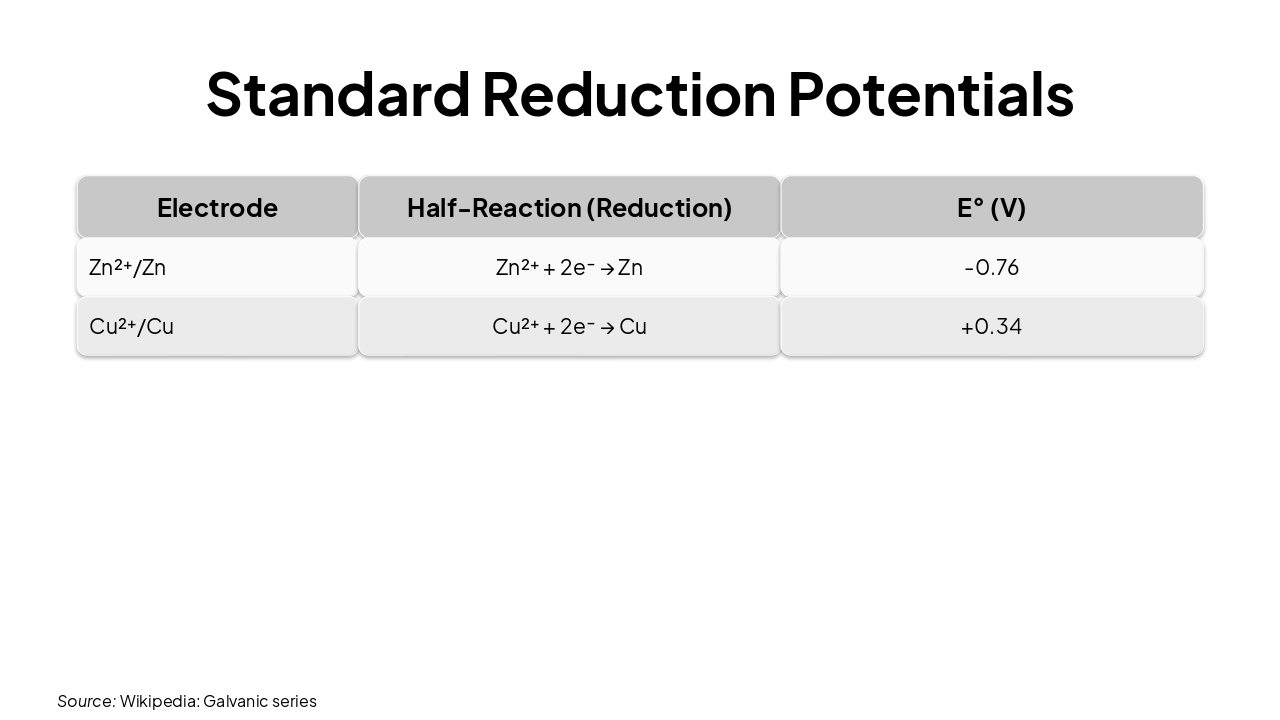
| Electrode | Half-Reaction (Reduction) | E° (V) |
|---|---|---|
| Zn²⁺/Zn | Zn²⁺ + 2e⁻ → Zn | -0.76 |
| Cu²⁺/Cu | Cu²⁺ + 2e⁻ → Cu | +0.34 |
Source: Wikipedia: Galvanic series
Source: Wikipedia: Galvanic series

Galvanic Cell: Spontaneous redox generates electricity Zinc-Copper Example:
Foundation of batteries and electrochemical applications
---
Photo by Jorge Campos on Unsplash

Explore thousands of AI-generated presentations for inspiration
Generate professional presentations in seconds with Karaf's AI. Customize this presentation or start from scratch.