Slide 1 - Chemical Bonds — Ionic & Covalent
Chemical Bonds — Ionic & Covalent
Adam, Tony, Jerson, Dillan Class Period:
---
Photo by Hal Gatewood on Unsplash

Generated from prompt:
SLIDE 1 — Names / Class Period / Topic Adam, Tony, Jerson, Dillan Class Period: ______ Topic: Chemical Bonds — Ionic & Covalent SLIDE 2 — Paragraph Explanation We chose the topic of chemical bonds because bonding is the foundation of all chemistry—every substance around us exists because atoms form bonds. Our favorite thing we learned was how ionic and covalent bonds behave differently, especially how electrons are transferred in ionic bonds but shared in covalent bonds. Understanding chemical bonding is important because it explains why materials have certain properties, how reactions occur, and how living organisms function at the molecular level. SLIDE 3 — Science Career Aligned With Topic Career: Chemical Engineer Degree Needed: Bachelor’s in Chemical Engineering Salary Range: $80,000–$150,000 per year Where They Work: Laboratories, manufacturing plants, oil & gas companies, pharmaceutical companies What They Do: Chemical engineers design processes that use chemical reactions and bonding principles to create products like medicines, plastics, fuels, and clean energy materials. SLIDE 4 — Key Vocabulary Atom: The smallest unit of matter. Valence Electrons: Electrons in the outermost shell involved in bonding. Ionic Bond: A bond formed when electrons are transferred from one atom to another. Covalent Bond: A bond formed when atoms share electrons. Electronegativity: An atom’s ability to attract electrons. Ion: A charged atom (cation = +, anion = –). SLIDE 5 — What Is a Chemical Bond? A chemical bond is a force that holds atoms together. Bonds form to make atoms more stable. Stability is achieved when atoms have a full outer electron shell. (Insert image: diagram of atoms bonding) SLIDE 6 — Ionic Bonds Formed between metals and nonmetals Electrons are transferred Creates positive (cation) and negative (anion) ions Strong attraction between opposite charges (Insert image: sodium + chlorine forming NaCl) SLIDE 7 — Properties of Ionic Compounds High melting and boiling points Conduct electricity when dissolved in water Form crystal lattice structures Usually solid at room temperature (Insert image: salt crystal lattice) SLIDE 8 — Covalent Bonds Formed between two nonmetals Electrons are shared Can form single, double, or triple bonds Molecules can be polar or nonpolar (Insert image: water molecule showing shared electrons) SLIDE 9 — Properties of Covalent Compounds Low melting and boiling points Do NOT conduct electricity Can be gases, liquids, or solids Often form molecules like CO₂, H₂O, O₂ (Insert image: CO₂ molecule) SLIDE 10 — Comparing Ionic vs Covalent Bonds Ionic Bonds: Transfer electrons Metal + nonmetal High melting point Conductive when dissolved Covalent Bonds: Share electrons Nonmetal + nonmetal Lower melting point Not conductive (Insert image: comparison chart) SLIDE 11 — Bibliography / Works Cited Khan Academy – Chemistry of Bonding ChemLibreTexts – Ionic & Covalent Bonds American Chemical Society – Chemical Bonding Overview SLIDE 12 — Pictures Include: Chemical engineer at work Ionic bond diagram (NaCl) Covalent bond diagram (H₂O, CO₂) Periodic table section showing metals vs nonmetals
This student presentation explores the fundamentals of chemical bonds, with a focus on ionic and covalent bonds. It explains why these bonds are foundational to chemistry, provides key vocabulary, details bond formation through electron transfer and sharing, compares properties of ionic and covalent compounds, and connects the topic to careers in chemical engineering. Features diagrams, tables, comparisons, and a bibliography.
Chemical Bonds — Ionic & Covalent
Adam, Tony, Jerson, Dillan Class Period:
---
Photo by Hal Gatewood on Unsplash


Source: Wikipedia Research: Chemical engineer

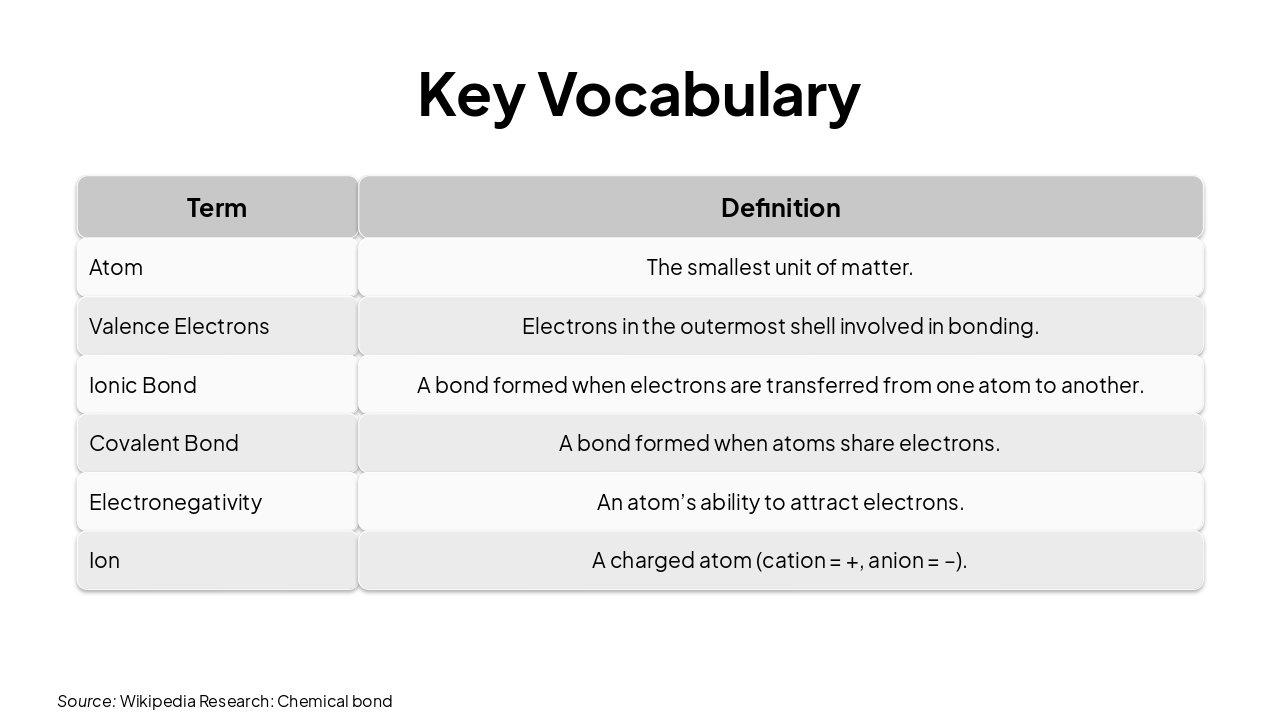
| Term | Definition |
|---|---|
| Atom | The smallest unit of matter. |
| Valence Electrons | Electrons in the outermost shell involved in bonding. |
| Ionic Bond | A bond formed when electrons are transferred from one atom to another. |
| Covalent Bond | A bond formed when atoms share electrons. |
| Electronegativity | An atom’s ability to attract electrons. |
| Ion | A charged atom (cation = +, anion = –). |
Source: Wikipedia Research: Chemical bond
---
Photo by Logan Voss on Unsplash
Source: Wikipedia Research: Chemical bond

---
Photo by Logan Voss on Unsplash
Source: Wikipedia Research: Chemical bond


---
Photo by Me Ollimono on Unsplash
Source: Wikipedia Research: Covalent bond


Ionic Bonds
Covalent Bonds



---
Photo by Ryan Zazueta on Unsplash
Source: Wikipedia Research: Chemical engineer



Explore thousands of AI-generated presentations for inspiration
Generate professional presentations in seconds with Karaf's AI. Customize this presentation or start from scratch.