Slide 1 - Stoichiometry of a Chemical Reaction
Stoichiometry of a Chemical Reaction
College of Natural and Computational Sciences Department of Chemistry
---
Photo by National Cancer Institute on Unsplash

Generated from prompt:
Make a presentation about
This educational presentation covers the fundamentals of chemical reaction stoichiometry. Key topics include writing and balancing chemical equations, classifying reactions such as acid-base, precipitation, and redox, mole-mole stoichiometry, limiting reactants and percent yield, and quantitative analysis methods like titration and gravimetric analysis. Designed for chemistry students in the College of Natural and Computational Sciences.
Stoichiometry of a Chemical Reaction
College of Natural and Computational Sciences Department of Chemistry
---
Photo by National Cancer Institute on Unsplash

---
Photo by Vinay Bhushan Meesala on Unsplash

4.1
From symbolic notation to balanced reactions per law of conservation of mass
---
Photo by Vitaly Gariev on Unsplash


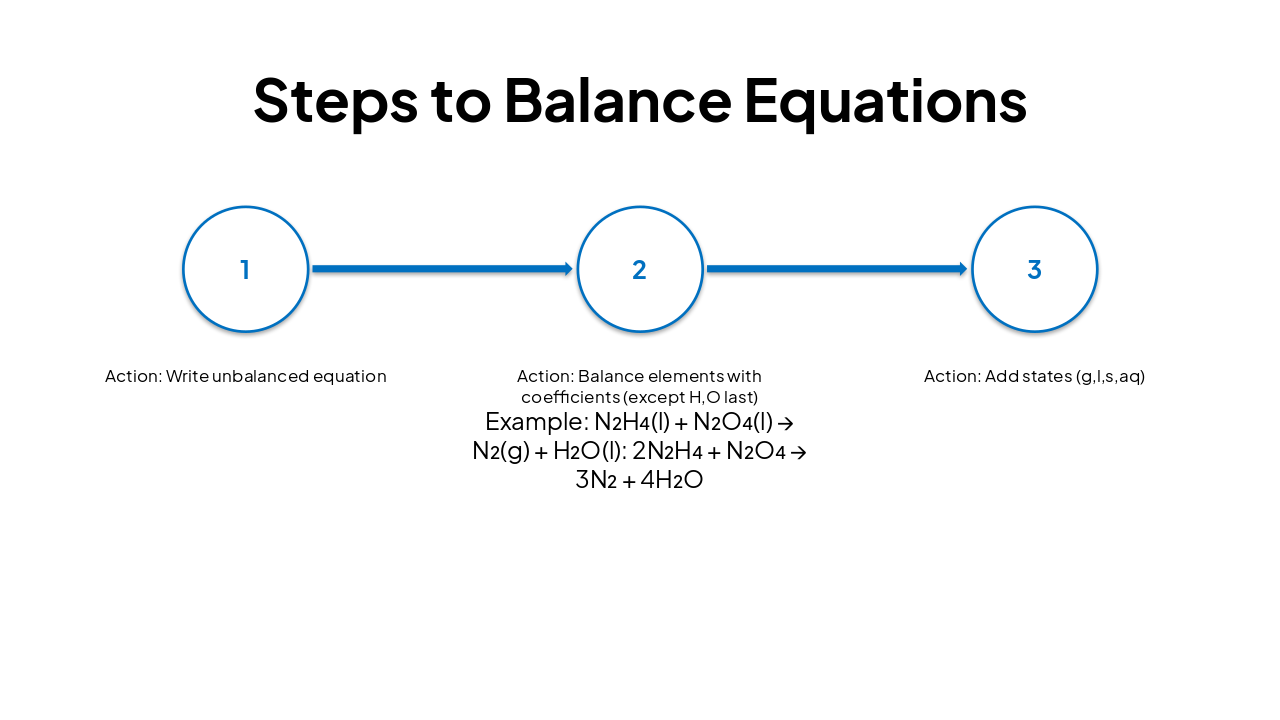
| Step | Action | Example: N₂H₄(l) + N₂O₄(l) → N₂(g) + H₂O(l) |
|---|---|---|
| 1 | Write unbalanced equation | |
| 2 | Balance elements with coefficients (except H,O last) | 2N₂H₄ + N₂O₄ → 3N₂ + 4H₂O |
| 3 | Add states (g,l,s,aq) |
Molecular Equation Ba(NO₃)₂(aq) + Na₂SO₄(aq) → BaSO₄(s) + 2NaNO₃(aq)
Net Ionic Equation Ba²⁺(aq) + SO₄²⁻(aq) → BaSO₄(s)
(Spectator ions: Na⁺, NO₃⁻ canceled from complete ionic)

4.2
Acid-base neutralization, precipitation, and oxidation-reduction reactions
---
Photo by Christian Kapeller on Unsplash


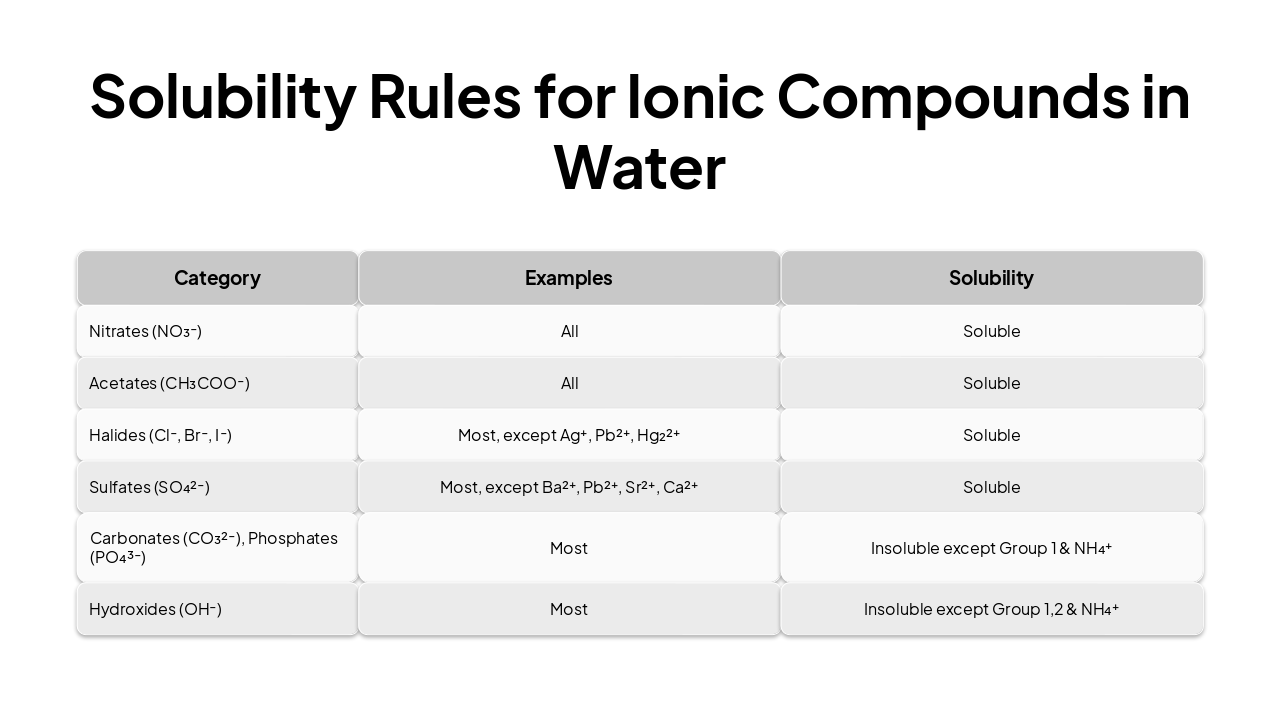
| Category | Examples | Solubility |
|---|---|---|
| Nitrates (NO₃⁻) | All | Soluble |
| Acetates (CH₃COO⁻) | All | Soluble |
| Halides (Cl⁻, Br⁻, I⁻) | Most, except Ag⁺, Pb²⁺, Hg₂²⁺ | Soluble |
| Sulfates (SO₄²⁻) | Most, except Ba²⁺, Pb²⁺, Sr²⁺, Ca²⁺ | Soluble |
| Carbonates (CO₃²⁻), Phosphates (PO₄³⁻) | Most | Insoluble except Group 1 & NH₄⁺ |
| Hydroxides (OH⁻) | Most | Insoluble except Group 1,2 & NH₄⁺ |

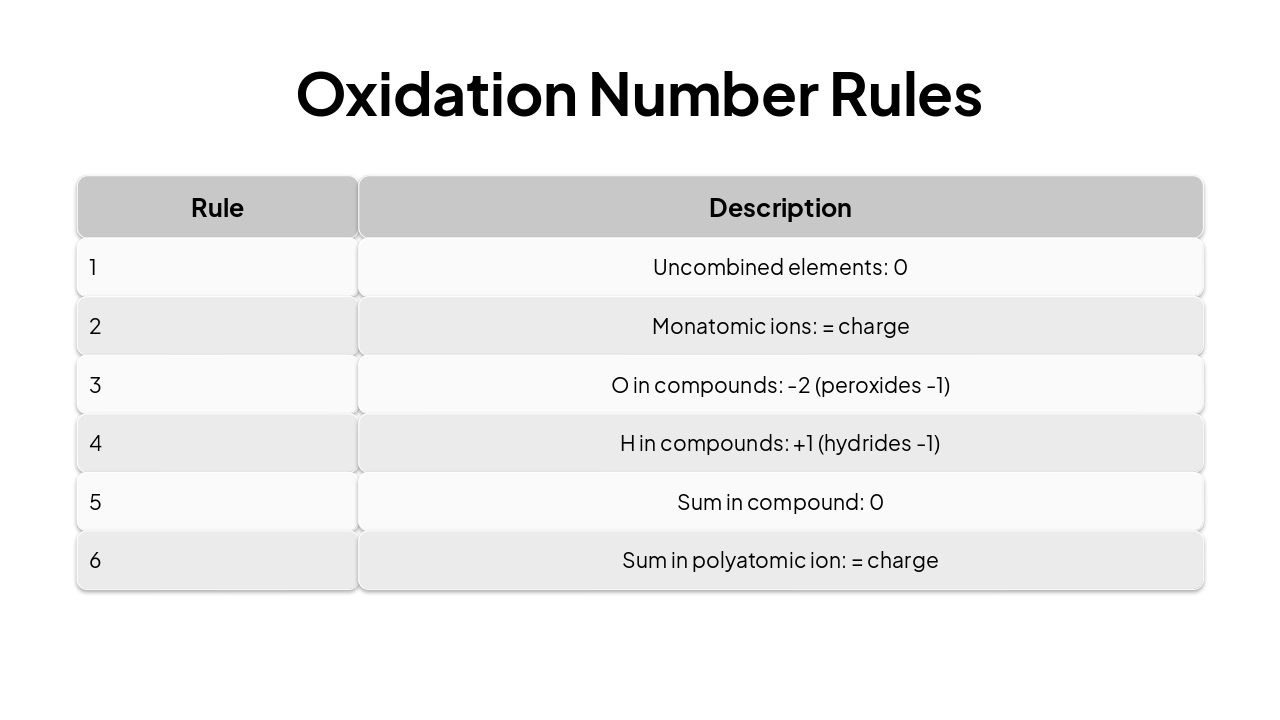
| Rule | Description |
|---|---|
| 1 | Uncombined elements: 0 |
| 2 | Monatomic ions: = charge |
| 3 | O in compounds: -2 (peroxides -1) |
| 4 | H in compounds: +1 (hydrides -1) |
| 5 | Sum in compound: 0 |
| 6 | Sum in polyatomic ion: = charge |
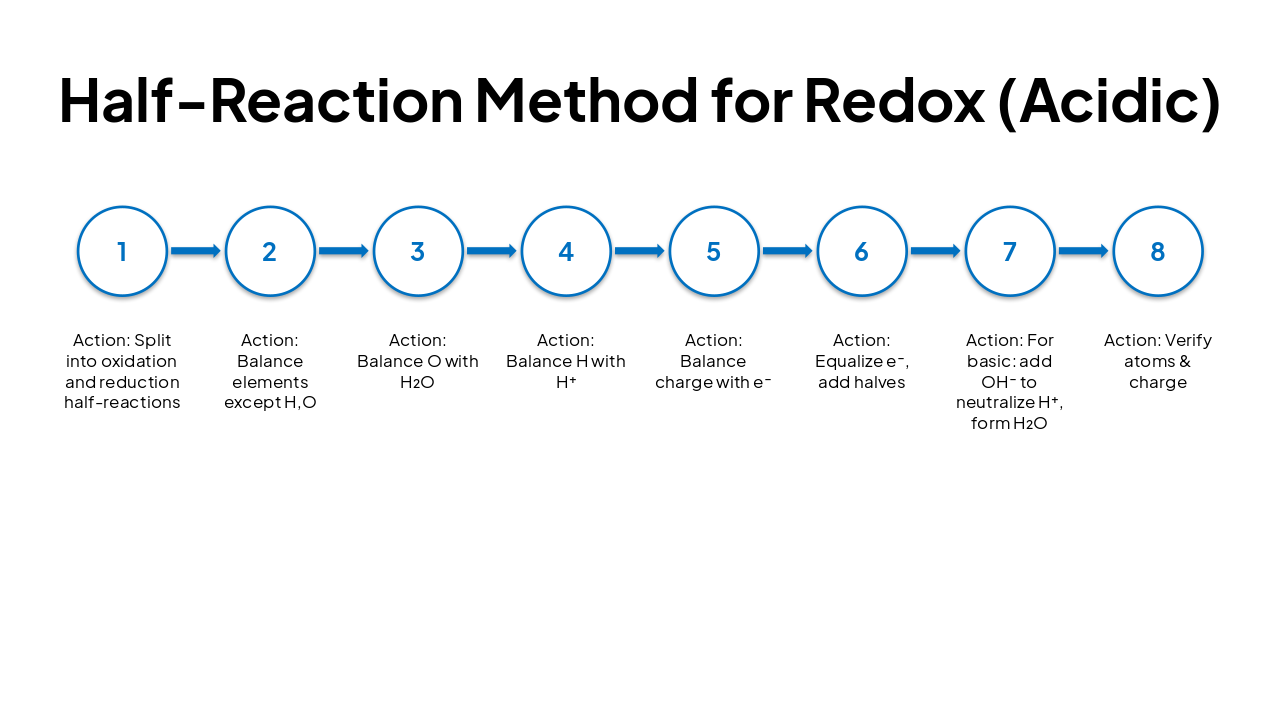
| Step | Action |
|---|---|
| 1 | Split into oxidation and reduction half-reactions |
| 2 | Balance elements except H,O |
| 3 | Balance O with H₂O |
| 4 | Balance H with H⁺ |
| 5 | Balance charge with e⁻ |
| 6 | Equalize e⁻, add halves |
| 7 | For basic: add OH⁻ to neutralize H⁺, form H₂O |
| 8 | Verify atoms & charge |
4.3
Mole-mole and mass-mole relationships in reactions
---
Photo by Myles Bloomfield on Unsplash

(2HCl + Ca → H₂ + CaCl₂; 1 mol H₂ / 2 mol HCl)

4.4
Limiting reactants and percent yield calculations
---
Photo by Stephan HK on Unsplash


---
Photo by Volodymyr Hryshchenko on Unsplash


Balanced chemical equations are key to stoichiometry: predict amounts, classify reactions, calculate yields, and perform analysis.
Thank you! Questions?
---
Photo by Omar:. Lopez-Rincon on Unsplash

Explore thousands of AI-generated presentations for inspiration
Generate professional presentations in seconds with Karaf's AI. Customize this presentation or start from scratch.