Slide 1 - Density Functional Theory
Density Functional Theory (DFT) Tutorial: Principles & Applications
A Comprehensive Undergraduate Tutorial | Presented by: [Lecturer Name]
---
Photo by Codioful (Formerly Gradienta) on Unsplash

Generated from prompt:
Improve and redesign a PowerPoint presentation on Density Functional Theory (DFT) for students. Make it modern, visually clean, and structured. Include: 1. Title slide (DFT tutorial, lecturer name placeholder) 2. Introduction to Computational Chemistry (simple, visual bullets) 3. What is Quantum Mechanics (simplified explanation) 4. Methods overview (Molecular Mechanics, Ab Initio, Semi-empirical, DFT, Molecular Dynamics) with comparison table 5. What is DFT (clear concept + diagram suggestion) 6. Theoretical models explanation 7. Software tools (Gaussian 09W, GaussView) with screenshots placeholders 8. Step-by-step: setting up calculations (job type, method, basis set) 9. Basis sets explained simply (6-31G*, 6-31+G*, etc.) with visual analogy 10. Practical selection rules (clean decision tree style) 11. Types of molecules and recommended methods (table) 12. Input/output files explanation 13. Visualization in GaussView (step-by-step simplified) 14. Frequency analysis (clear visuals: stable vs transition state) 15. Thermochemical outputs (ZPE, H, S, G) simplified 16. HOMO-LUMO concept (diagram + equation) 17. HOMO-LUMO gap and interpretation 18. Molecular Electrostatic Potential (MEP) with color legend 19. Global reactivity parameters (simplified formulas + meaning) 20. UV-Vis & TD-DFT explanation 21. Setting up TD-DFT calculations 22. Output interpretation (tables and visuals) 23. Summary slide (key takeaways) 24. Thank you / Q&A slide Design instructions: - Use minimal text per slide - Add icons/visual placeholders - Use consistent color theme (blue/purple scientific theme) - Add section divider slides - Improve grammar and clarity throughout - Convert dense text into bullet points and diagrams Make it suitable for undergraduate teaching and easy to follow.
Comprehensive undergraduate tutorial on Density Functional Theory (DFT) in computational chemistry. Covers introduction to quantum chemistry, theoretical methods overview, DFT principles, practical workflows with Gaussian/GaussView, and analysis of H
Density Functional Theory (DFT) Tutorial: Principles & Applications
A Comprehensive Undergraduate Tutorial | Presented by: [Lecturer Name]
---
Photo by Codioful (Formerly Gradienta) on Unsplash

---
Photo by Codioful (Formerly Gradienta) on Unsplash

1
Bridging Theory and Experimentation
---
Photo by Salvus on Unsplash

---
Photo by Drew Beamer on Unsplash

---
Photo by Drew Beamer on Unsplash

2
Choosing the Right Tool for the Job
---
Photo by Salvus on Unsplash

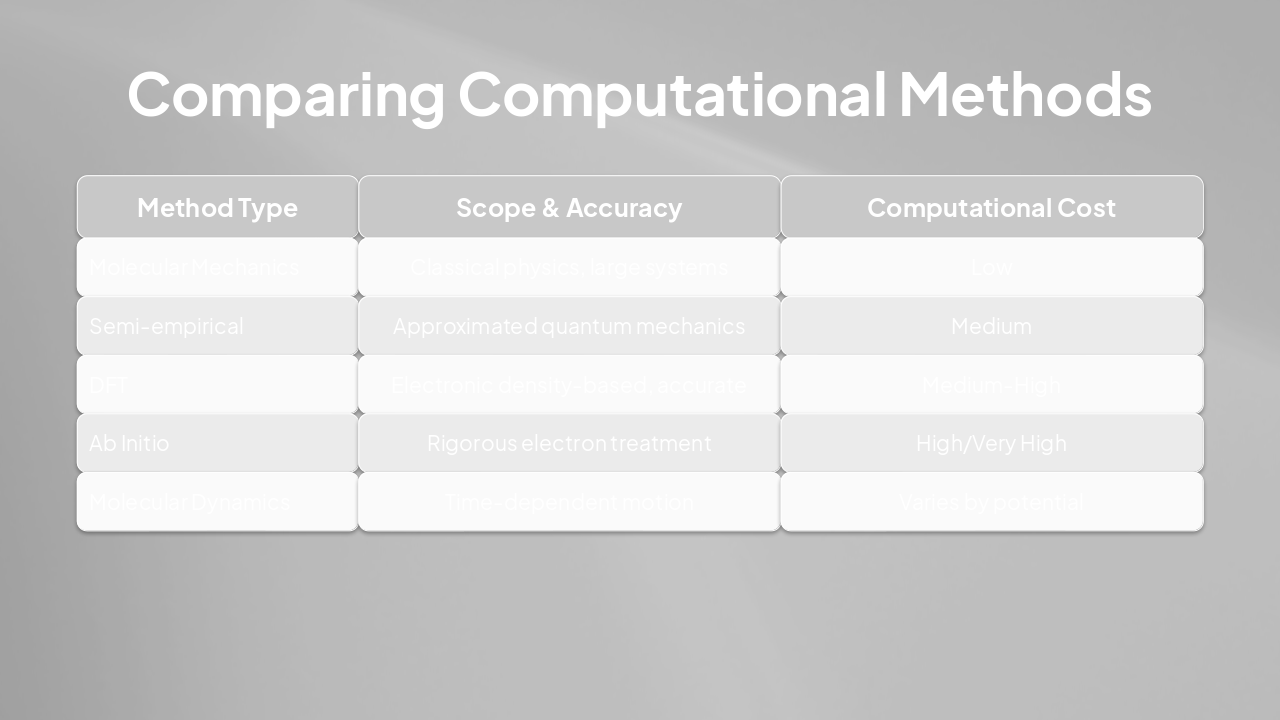
| Method Type | Scope & Accuracy | Computational Cost |
|---|---|---|
| Molecular Mechanics | Classical physics, large systems | Low |
| Semi-empirical | Approximated quantum mechanics | Medium |
| DFT | Electronic density-based, accurate | Medium-High |
| Ab Initio | Rigorous electron treatment | High/Very High |
| Molecular Dynamics | Time-dependent motion | Varies by potential |
---
Photo by Drew Beamer on Unsplash
3
Principles and Models
---
Photo by Salvus on Unsplash

---
Photo by Drew Beamer on Unsplash

---
Photo by Drew Beamer on Unsplash

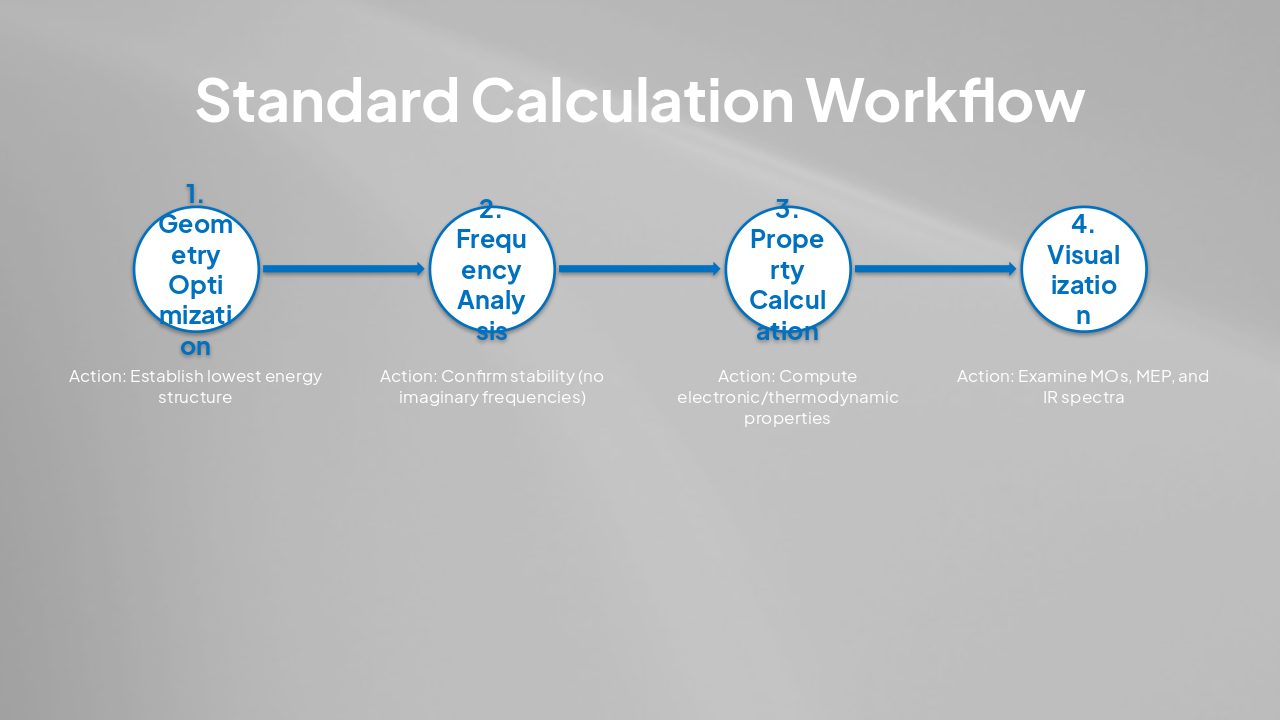
| Step | Action |
|---|---|
| 1. Geometry Optimization | Establish lowest energy structure |
| 2. Frequency Analysis | Confirm stability (no imaginary frequencies) |
| 3. Property Calculation | Compute electronic/thermodynamic properties |
| 4. Visualization | Examine MOs, MEP, and IR spectra |
---
Photo by Drew Beamer on Unsplash
5
Extracting Meaning from Data
---
Photo by Salvus on Unsplash

---
Photo by Drew Beamer on Unsplash Photo by Norbert Kowalczyk on Unsplash

---
Photo by Drew Beamer on Unsplash

Thank you! Questions?
DFT enables deep insights into chemical phenomena through efficient computational models. Keep practising with Gaussian and GaussView to master the workflow.
---
Photo by Codioful (Formerly Gradienta) on Unsplash

Explore thousands of AI-generated presentations for inspiration
Generate professional presentations in seconds with Karaf's AI. Customize this presentation or start from scratch.