Slide 1 - Title Slide
The New Drug Development Cycle
Understanding the Stages, Incentives, and Regulatory Requirements in Pharmaceutical Development

Generated from prompt:
Create a detailed PowerPoint presentation based on the uploaded document 'New Drug Development Cycle'. Slides should cover: overview (cost, timeline, success rates), Pre‑Discovery/Drug Discovery methods and incentives (NIH grants, SBIR/STTR, Orphan Drug Act), Pre‑Clinical stage (IACUC approval, in vitro vs in vivo testing, pharmacology & toxicology, GLP principles, dosage formulation, ADME studies, GMP manufacturing and the 5 P’s, IND application components), Clinical Development (trial protocol, participant selection, Phases 1–3 with sample sizes and goals, FDA expedited programs like Fast Track, Breakthrough Therapy, Accelerated Approval, Priority Review, and orphan drug incentives), FDA Approval process (NDA/BLA submission, review categories, Hatch‑Waxman Act, patent term extension), and Post‑Marketing Monitoring (FAERS and safety surveillance). Include clear diagrams or process flow slides and a final summary slide.
Explore the pharmaceutical drug development lifecycle, from discovery and pre-clinical testing to clinical trials, FDA approvals, expedited pathways, and post-market surveillance. Covers costs, success rates, funding incentives like SBIR/STTR and the
The New Drug Development Cycle
Understanding the Stages, Incentives, and Regulatory Requirements in Pharmaceutical Development



---
Photo by Logan Voss on Unsplash

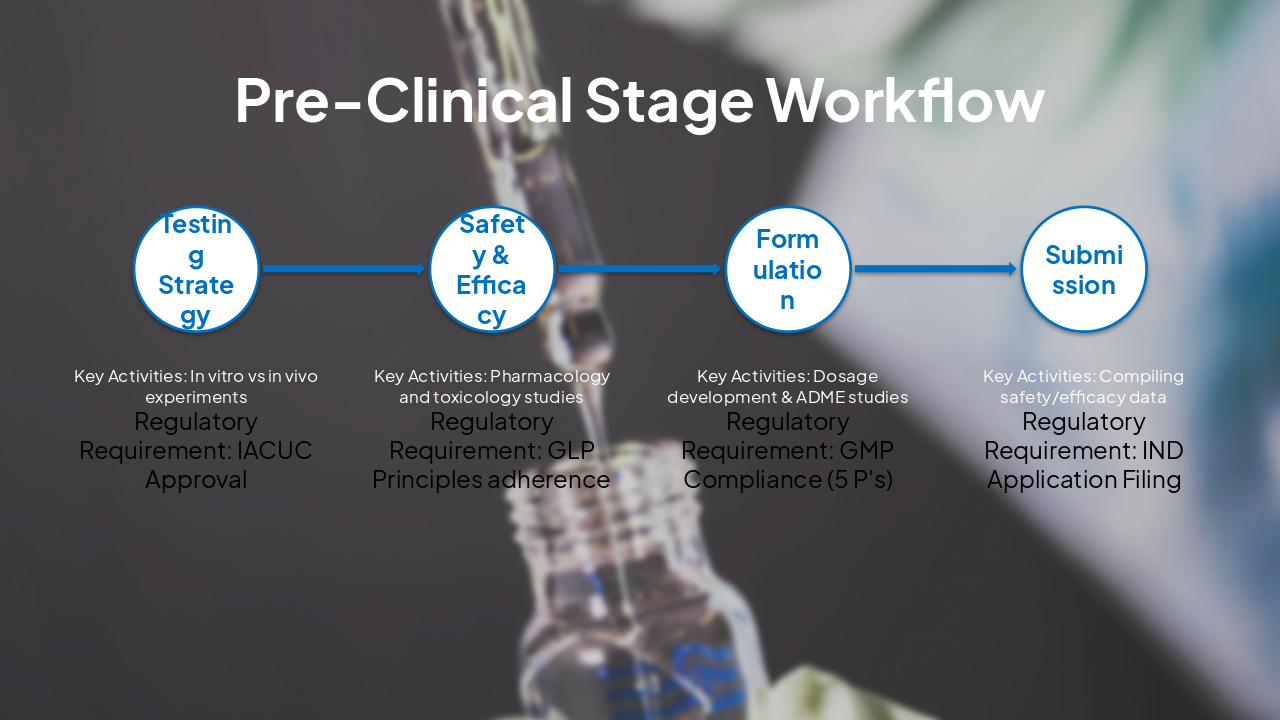
| Step | Key Activities | Regulatory Requirement |
|---|---|---|
| Testing Strategy | In vitro vs in vivo experiments | IACUC Approval |
| Safety & Efficacy | Pharmacology and toxicology studies | GLP Principles adherence |
| Formulation | Dosage development & ADME studies | GMP Compliance (5 P's) |
| Submission | Compiling safety/efficacy data | IND Application Filing |
---
Photo by CDC on Unsplash
Phase 1: Clinical Trials Part 1 Small group (20-100), focusing on safety and dosage. Phase 2: Clinical Trials Part 2 Medium group (100-500), testing efficacy and side effects. Phase 3: Clinical Trials Part 3 Large group (1,000-5,000+), verifying efficacy against standard treatments.



---
Photo by Tim Cooper on Unsplash

Summary: Navigating the Drug Development Lifecycle
The journey from lab bench to patient bedside is complex, costly, and heavily regulated. Success requires scientific rigor, strategic funding, and alignment with FDA pathways.

Explore thousands of AI-generated presentations for inspiration
Generate professional presentations in seconds with Karaf's AI. Customize this presentation or start from scratch.